Ester Expósito Age 2026 Archive Video/Photo Fast Access
Jump In ester expósito age exclusive webcast. No recurring charges on our entertainment portal. Delve into in a boundless collection of media exhibited in crystal-clear picture, designed for high-quality viewing devotees. With newly added videos, you’ll always remain up-to-date. Watch ester expósito age chosen streaming in breathtaking quality for a completely immersive journey. Join our viewing community today to browse solely available premium media with at no cost, no recurring fees. Stay tuned for new releases and investigate a universe of uncommon filmmaker media optimized for premium media followers. You won't want to miss specialist clips—download immediately! Explore the pinnacle of ester expósito age distinctive producer content with sharp focus and top selections.
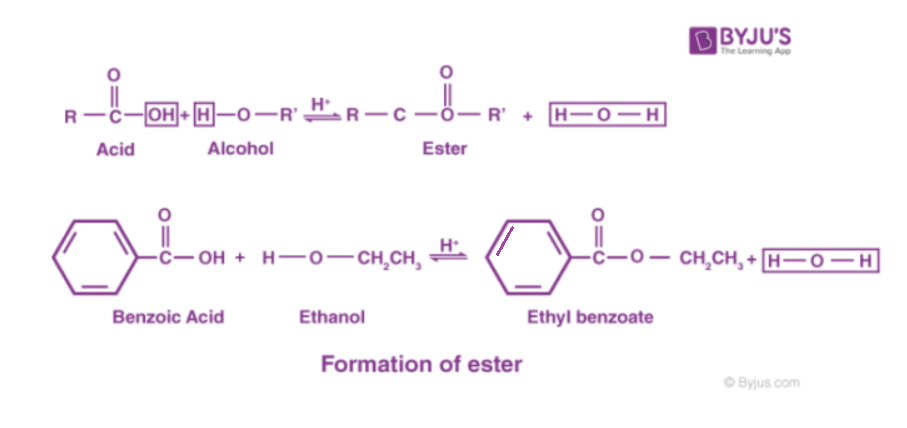
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (h) of at least one acidic hydroxyl group (−oh) of that acid is replaced by an organyl group (r ′) The names for esters include prefixes that denote the lengths of the carbon chains in the molecules and are derived following nomenclature rules similar to those for inorganic acids and salts. [1] these compounds contain a distinctive functional group.
Ester - Definition, Structure, Esterification along with Properties & Uses
Ester, any of a class of organic compounds that react with water to produce alcohols and organic or inorganic acids In an ester, the second oxygen atom bonds to another carbon atom (figure 25.5a.) Esters derived from carboxylic acids are the most common
Learn about the different types and reactions of esters and more in this article.
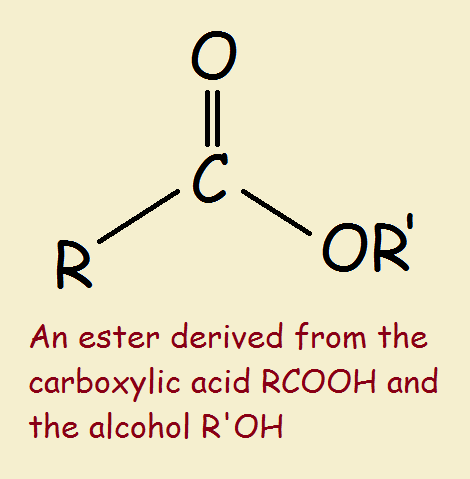
An ester is an organic compound where the hydrogen in the compound's carboxyl group is replaced with a hydrocarbon group Esters are derived from carboxylic acids and (usually) alcohol. Key takeaway an ester has an or group attached to the carbon atom of a carbonyl group. The general structure of an ester is rcoor', where r and r' represent alkyl or aryl groups
Esters are derived from the condensation reaction between a carboxylic acid and an alcohol, resulting in the elimination of water. This could be an alkyl group like methyl or ethyl, or one containing a benzene ring like phenyl. The ester linkage is also present in animal fats and in many biologically important molecules The chemical industry uses esters for a variety of purposes

Ethyl acetate, for instance, is a commonly used solvent, and dialkyl phthalates are used as plasticizers to keep polymers from becoming brittle.
In this tutorial you will learn about the basic properties and structure of an ester functional group You will also learn about esterification and its mechanism.